
What is BMC?
BMC (Bone Marrow Aspirate Concentrate) is a cell concentrate derived from bone marrow aspirate, called bone marrow aspirate concentrate.
Bone marrow is a fluid tissue rich in a heterogeneous population of mature cells and multipotent cells. Among them are the mesenchymal stromal cells (MSC), which are the precursor cells of musculoskeletal tissue.
RegenKit Extracell BMC kits are specifically manufactured for the effective processing of bone marrow aspirate (BMA), through a short centrifugation in RegenTHT tubes.
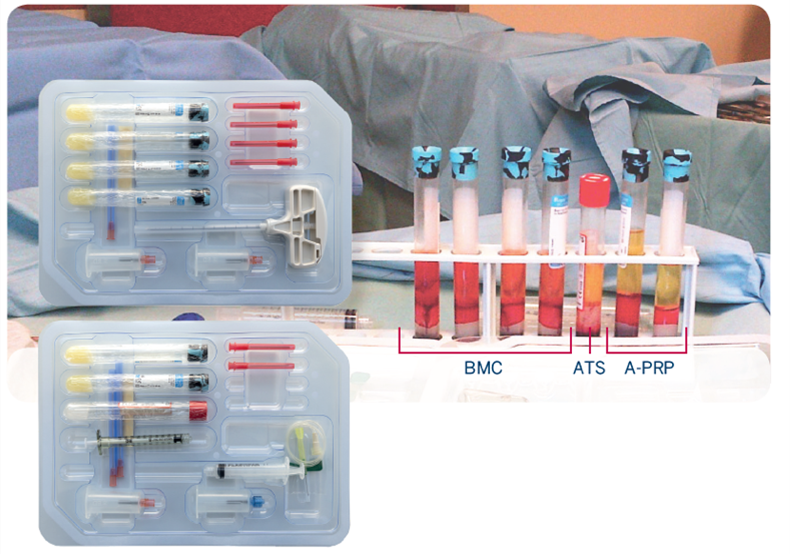
These tubes contain a biologically inert separation gel with a specific gravity that stabilises the physical separation of plasma and mononuclear cell (MNC) fractions from the other cellular elements of the bone marrow aspirate. This allows intraoperative processing of bone marrow aspirate to produce a bone marrow aspirate concentrate that is reimplanted during the same surgical procedure without the need for cell selection and expansion in the laboratory (‘one-step therapy’) (Holton et al., 2016).More than 80% of MSCs are recovered in the resulting cell concentrate. In vitro, these cells form colony-forming fibroblast units (CFU-F) capable of undergoing osteogenesis, chondrogenesis and adipogenesis when cultured in their respective specific induction medium. By transferring all the regenerative potential in bone marrow aspirate to the site of the lesion, bone marrow aspirate concentrate represents an interesting therapeutic option for treatment.

Please always read the operating instructions before use:
Preparation of autologous platelet-rich plasma and other plasma-based products.
Class IIb CE certified medical devices
Regen Lab SA is a medical device manufacturer certified to ISO13485 :2016 and MDSAP.
CE2797
Warnings and precautions:
Sterility must be maintained throughout the phlebotomy procedure. Take adequate precautions to avoid contact with the patient’s blood or cross-contamination. Take adequate precautions to protect against needles or broken glass tubes. Do not use any of the sterile components of the kit if the packaging has been opened or damaged. Do not use any component of this kit that is damaged or broken. Do not use the tube if it has lost vacuum. Do not use the sodium citrate or other components of the tube alone. Store between 5ºC and 30ºC; bring the kit to room temperature before use. Do not use after the expiry date.
Disposable; do not reuse any part of the kit. Reuse may cause infection or other
illness/injury.
Transfer needles must only be used for fluid transfer or preparation of the injection, but not for the injection itself. Preparation of platelet rich plasma (PRP) must be performed by or under the supervision of a physician trained in the device and procedures. PRP preparation must only be used by a doctor trained in the procedures. The patient must be informed of general risks and possible adverse effects. The platelet-rich plasma (PRP) must be prepared with fresh blood and used within four hours (product, use as soon as possible and do not
store). Dispose of the tubes and other components after use according to the disposal guidelines for potentially contaminated blood products.
Use a centrifuge with swing-out or fixed-angle rotor (e.g. RegenPRP Centri from Regen Lab). Follow the manufacturer’s instructions when using the centrifuge. The tubes should be centrifuged at 1500 RZB as described in the instructions for use. High centrifugal forces (greater than 2200 RZB) may cause the tubes to break, come into contact with the blood or possibly cause injury. A lower centrifugal force (less than 1500 RZB) may result in insufficient separation of the blood and contamination of the PRP with red blood cells. The centrifuge inserts should specifically match the size of the tubes. The use of inserts that are too small or too large may cause the tubes to break. Care should be taken to ensure that the tubes are properly seated in the holder in the centrifuge. The tubes must be balanced in the rotor to
avoid possible breakage of the glass.
Possible undesirable side effects:
Injury to blood vessels, haematoma, delayed wound healing and/or infection. Temporary or permanent damage to nerves which may result in pain or numbness. Early or late surgical infection.
→ Contraindications and side effects according to IFU


